Medical Science
Medical Needs Are Learned on Site.
Offering True Value with Our Pharmaceutical
Products
We proactively conduct various activities to accurately grasp the needs of patients,
their families and healthcare professionals to execute medical communication and provision of medical
information to address those needs in a scientifically objective, unbiased, reliable, and evidence-based
manner.
The value of pharmaceuticals increases as post-marketing information (evidence) is accumulated
and added to the compound information. The department constantly makes efforts to determine medical needs that
are not adequately met for patients, their families and healthcare professionals, and engages in various
evidence creating activities to fulfill these unmet needs. We believe that information based on the evidence
we generate is essential because it will serve as a reassuring factor for healthcare professionals and help
ensure appropriate treatment for the patients.
Medical Affairs Function
The Medical Affairs function operates based on voluntary standards issued by the Japan Pharmaceutical Manufacturers Association (JPMA)*1, such as “Consensus Statement on Medical Affairs Activities” and “Consensus Statement on Medical Science Liaison Activities.” To ensure that activities are independent of marketing and not conducted for the purpose of gaining sales of our pharmaceutical products, we endeavor to maximize their medical value for patients and healthcare professionals and promote their proper use. This is done by creating evidence through advanced medical and scientific exchanges with healthcare professionals and disseminating information based on it. Medical Science Liaison (MSL) strives to identify medical needs that lead to creating new evidence, additional formulations and expanding indications through scientific discussion with healthcare professionals, and provide high quality information in response to inquiries from them.
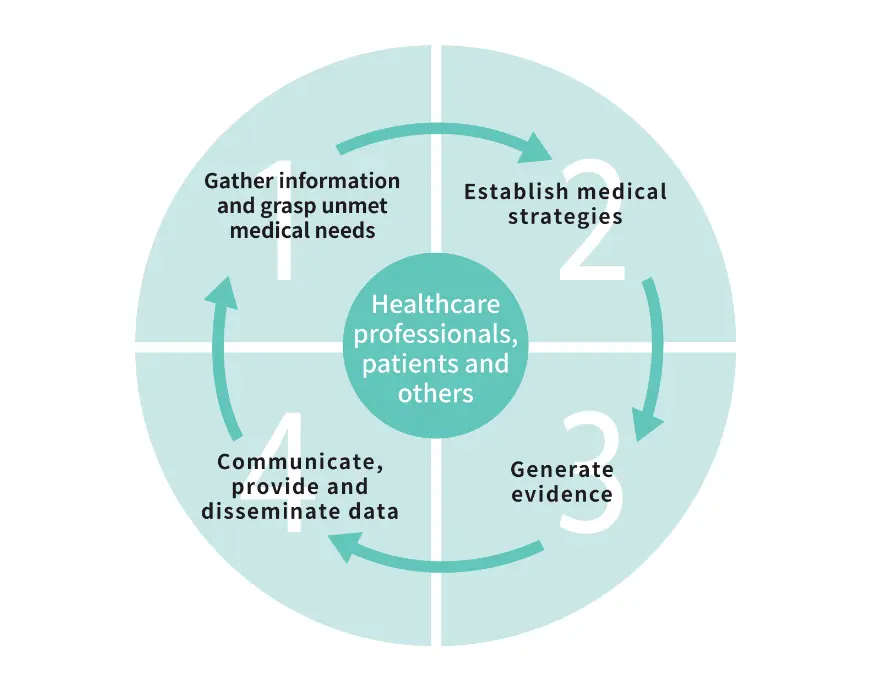
We are currently making efforts to promote medical activities such as evidence generation, provision, and dissemination primarily in the areas of psychiatry & neurology, diabetes, and oncology. The research outcomes obtained through evidence generation activities are disseminated to healthcare professionals through conference presentations and publication of papers*2. Additionally, we are actively engaged in database studies utilizing real-world data, which is widely utilized worldwide. By transparently publishing the results of these studies, we aim to contribute to the daily medical practices of healthcare professionals. Achieving the SDGs requires governments and other decision-makers to have access to necessary resources in all regions of the world and to invest their full efforts into scientific and technological innovations. We believe that by openly publishing research findings as open access papers as far as possible, we can contribute to the achievement of the SDGs.
- *2Sumitomo Pharma Medical Affairs Website for healthcare professionals: https://sumitomo-pharma.jp/medical_affairs/ (in Japanese only)
Medical Information Function
In the Medical Information function, we produce materials for patients to promote the proper use of our pharmaceutical products and support the provision of product information based on scientific evidence to healthcare professionals by our medical representatives (MRs). We also create informational materials such as “Kusuri-no-shiori”, (drug information sheets) and “Drug Guides for Patients,” which healthcare professionals can use to explain to patients. These materials contain details on dosage instructions and specific symptoms of adverse reactions. In order to provide 24-hour support for community healthcare in the future, we will disseminate easy-to-understand information through our websites directed at patients and their families, and healthcare professionals, that meets their various needs.
Product Information System
Pharmaceuticals can be effective when used along with information on their efficacy and safety. We believe that the prompt and accurate provision of such information will contribute to maximizing the medical value of our products for patients and healthcare professionals. Therefore, in addition to information provided on our website, we strive to promptly and accurately respond to inquiries from healthcare professionals through our MRs and Product Information Center using in-house product information system, by reviewing and supplementing the content with an awareness of the needs of healthcare professionals, we will strive to further enhance the reliability of the information provided as well as customer satisfaction.
Act as an Information Hub Further Utilizing Customer Feedback
As a contact point related to our products, we have established the "Product Information Center " to respond to inquiries from patients, their families, and healthcare professionals. We aim to continue providing prompt, accurate, and courteous information on proper usage, contributing to the well-being of patients. Additionally, we will strengthen our role of analyzing customer feedback obtained through inquiry responses, appropriately relaying it within the organization, and visualizing trends in customer feedback to drive improvements.
Total number of inquiries in fiscal 2023 (April 1, 2023 - March 31, 2024) was approximately 25,800.
For more information on the Product Information Center including total numbers of inquiries received, please see “ESG Data Table.”
As a research and development-oriented pharmaceutical company with a global presence, Sumitomo Pharma aims to continuously create innovative and effective medicines in areas with high unmet medical needs. Through the provision of high quality information on our products created, we will make efforts to further enhance our contribution to the betterment of healthcare and fuller lives of people worldwide.